|
Let us study what is an amide and the amide nomenclature. It's made from ammonia and carbon dioxide in large quantities for use in fertilisers, animal feed, and the production of urea-formaldehyde resins, which are used to make plastics. Urea, also known as carbamide, is a crystalline compound produced as a byproduct of protein synthesis and removed in the urine of mammals. Imides are compounds having two carbonyl (CO) groups linked to the same nitrogen atom created by reacting amides with acid chlorides or anhydrides.Īcetamide, also known as ethanamide (CH 3 CONH 2 ), and dimethylformamide HCON(CH 3 ) 2, which are used as solvents, sulfa drugs, and nylons, are among the commercially important amides. Lithium aluminium hydride, a powerful reducing agent, converts amides to amines. Amides are difficult to oxidise or reduce, however hydrogenation (the addition of hydrogen at high temperatures and pressures) will convert most carboxylic acid amides to amines in the presence of a catalyst. Amides can also be dehydrated to produce nitriles. Hydrolysis (a chemical reaction with water) is the reaction by which covalent amides are changed to acids and amines this reaction is usually slow unless it is catalysed by a strong acid, an alkali, or an enzyme. They can also be created when water reacts with nitriles. Simple amides are usually created by reacting acids or acid halides with ammonia or amines. They are nonconductors of electricity and are both organic and inorganic solvents.Even covalent amides with low molecular weight have high boiling points.Īlthough polyamides (amides joined together to create huge molecules called polymers) are abundant as the protein of living systems, there are no practicable natural sources of simple covalent amides. With the exception of formamide, which is a liquid, covalent amides produced from ammonia are solids those with fewer than five carbon atoms are water soluble. 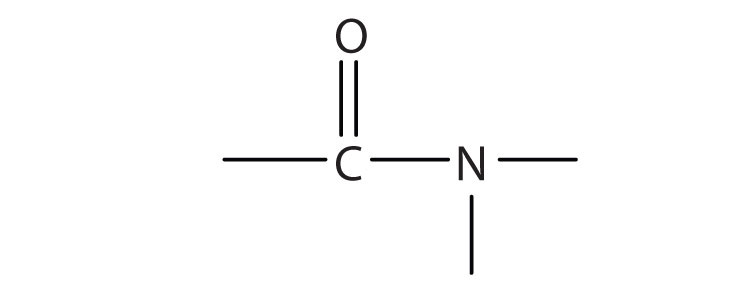
The amide formula or the amide group formula is CO-NH. Ionic amides, also known as saltlike amides, are extremely alkaline compounds made by reacting ammonia, an amine, or a covalent amide with a reactive metal such as sodium. Sulfonamides (RSO 2 NR 2 ) are related to sulfonic acids in structure (RSO 3 H). The most important group is carboxamides (R′CONR 2 ), which are derived from carboxylic acids (R′COOH). When the hydroxyl group (OH) of an acid is replaced by an amino group, the result is a neutral or very weakly acidic compound (NR2, in which R may represent either as a hydrogen atom or an organic combining group, which is such as methyl, CH 3 ). 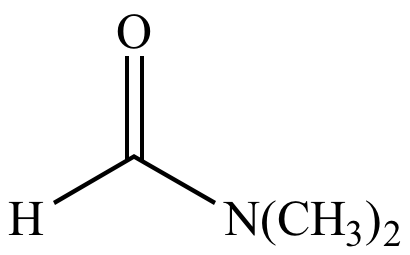
Amide is a nitrogen-containing compound that belongs to one of two classes: ammonia and amines.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
